by Eileen Luders, Nicolas Cherbuin, Christian Gaser: : Younger Brains In Long-Term Meditation Practitioners…
Normal aging is known to be accompanied by loss of brain substance. The present study was designed to examine whether the practice of meditation is associated with a reduced brain age. Specific focus was directed at age fifty and beyond, as mid-life is a time when aging processes are known to become more prominent. We applied a recently developed machine learning algorithm trained to identify anatomical correlates of age in the brain trans- lating those into one single score: the BrainAGE index (in years). Using this validated approach based on high- dimensional pattern recognition, we re-analyzed a large sample of 50 long-term meditators and 50 control subjects estimating and comparing their brain ages. We observed that, at age fifty, brains of meditators were es- timated to be 7.5 years younger than those of controls. In addition, we examined if the brain age estimates change with increasing age. While brain age estimates varied only little in controls, significant changes were detected in meditators: for every additional year over fifty, meditators’ brains were estimated to be an additional 1 month and 22 days younger than their chronological age. Altogether, these findings seem to suggest that meditation is beneficial for brain preservation, effectively protecting against age-related atrophy with a consistently slower rate of brain aging throughout life.
© 2016 Elsevier Inc. All rights reserved.
1. Introduction
Meditation is attracting increasing interest in relation to health and wellbeing but its biological effects are not well understood. Although publication bias and methodological limitations are strong concerns in this emerging field, meditation has been shown to induce increases in brain tissue, even after relatively short periods of time, such as weeks or months (Fox et al., 2014). Complementing these short-term effects, brains of long-term meditators have been reported to be structurally different, with thicker, better connected, and more complex cortical sec- tions, larger volumes, areas and dimensions of specific brain regions, as well as more local brain tissue than in brains of healthy controls (Luders et al., 2013a, 2015; Fox et al., 2014).
Since normal aging is known to be accompanied by loss of brain sub- stance (Raz et al., 2010; Pfefferbaum et al., 2013), the question arises as to whether meditation may have a protective effect against age-related brain atrophy, where effects might accumulate over time and become evident especially in later years of life. The present study was designed to examine whether the practice of meditation manifests as a seemingly reduced brain age in meditators. Specific focus was directed at age fifty and beyond, as mid-life is a time when aging processes are known to be- come more salient as well as more functionally significant (Fraser et al., 2015).
We utilized a recently developed and validated high-dimensional pattern recognition approach which allows estimating, automatically and objectively, the age of any given brain based on a single T1- weighted brain image (Franke et al., 2010, 2012). Importantly, in people aged 19–86 years, this method has been shown to predict brain age with a mean error of as little as 4.98 years (Franke et al., 2010). Moreover, as also evaluated previously (Franke et al., 2010), the 95% confidence in- terval for the prediction of brain age is stable across the entire age range, even in older adults (e.g., age [mean ± SD] = 20 ± 11.6 years vs. age [mean ± SD] = 80 ± 11.7 years). This is especially relevant for our current sample, which is composed of 100 subjects (50 medita- tors/50 controls) ranging in age between 24 and 77 years.
The applied method effectively translates the complex, multidimen- sional aging pattern across the whole brain into one single score: the brain age (BrainAGE) index. The polarity of the index indicates if brains appear younger (negative score) or older (positive score) than their chronological age, and the numeric value specifies the magnitude of the difference (in years) between estimated age and chronological age. For example, estimating the brain ages of individuals with mild Alzheimer’s disease in a previous study revealed a mean BrainAGE.
⁎ Corresponding author at: Department of Neurology, UCLA School of Medicine, 635 Charles E Young Drive South, Suite 225, Los Angeles, CA 90095-7334, USA.
E-mail address: eileen.luders@ucla.edu (E. Luders). 1 Both authors contributed equally.
http://dx.doi.org/10.1016/j.neuroimage.2016.04.007
1053-8119/© 2016 Elsevier Inc. All rights reserved.
index of +10 (Franke et al., 2010). These striking findings suggested significantly accelerated brain aging in Alzheimer’s patients compared to normal aging in healthy controls (Franke et al., 2010). In contrast, for the current study focusing on the potential brain-preserving effect of long-term meditation, we predicted decelerated brain aging in med- itators compared to normal aging in healthy controls. With respect to the magnitude of the effect, the deviation from ‘normality’ may range between a few days to several years.
2. Methods
2.1. Subjects
Note that this was a re-analysis of data overlapping, fully or in part, with those used in prior studies (Luders et al., 2009, 2011, 2012a,b, 2013a,b, 2014, 2015; Kurth et al., 2015a,b). The study sample included 50 meditation practitioners (28 men, 22 women) and 50 control sub- jects (28 men, 22 women). Meditators and controls were closely matched for chronological age, ranging between 24 and 77 years with a mean age of 51.4 years in both groups (SD = 12.8 years in meditators; SD = 11.8 years in controls). Meditators were recruited from various venues in the greater Los Angeles area and had close to twenty years of meditation experience on average (mean: 19.8 years; SD = 11.4 years; range: 4–46 years). A detailed overview with respect to each subject’s individual practice has been previously provided (Luders et al., 2012a). Brain scans for the control subjects were obtained from the ICBM database of normal adults (http://www.loni.usc.edu/ ICBM/Databases/). All subjects gave their informed consent in accor- dance with the policies and procedures of UCLA’s Institutional Review Board.
2.2. Image Acquisition
All subjects were scanned at the same site, using the same scanner, and following the same scanning protocol. Specifically, magnetic reso- nance images were acquired on a 1.5 Tesla Siemens Sonata scanner (Erlangen, Germany) using an 8-channel head coil and a T1-weighted magnetization-prepared rapid acquisition gradient echo sequence with the following parameters: 1900 ms repetition time, 4.38 ms echo time, 15° flip angle, 160 contiguous sagittal slices, 256 × 256 mm2 field-of-view, and 1 × 1 × 1 mm3 voxel size.
2.3. Data Preprocessing
All T1-weighted images were processed in Matlab (http://www. mathworks.com/products/matlab/) utilizing SPM8 (http://www.fil.ion. ucl.ac.uk/spm) and the VBM8 toolbox (http://dbm.neuro.uni-jena.de/ vbm.html), as recently described (Gaser et al., 2013). Briefly, images were bias corrected, spatially normalized, and classified into gray matter, white matter, and cerebrospinal fluid, all within the same gen- erative model (Ashburner and Friston, 2005). The segmentation proce- dure was extended by accounting for partial volume effects (Tohka et al., 2004), applying adaptive maximum a posteriori estimations (Rajapakse et al., 1997), and using a hidden Markov Random Field model (Cuadra et al., 2005). The resulting gray matter partitions were then smoothed using an 8 mm full-width-at-half-maximum (FWHM) Gaussian kernel. Subsequently, image resolution was set to 8 mm, and further data reduction was performed via principal component analysis utilizing the “Matlab Toolbox for Dimensionality Reduction” (http:// lvdmaaten.github.io/drtoolbox/), using the maximal number of princi- pal components that equals to the size of the training sample, minus 1. Finally, using the gray matter data, the individual brain ages were predicted utilizing “Spider for Matlab” (http://www.kyb.mpg.de/bs/ people/spider/main.html). For this purpose, we leveraged a recently developed BrainAGE estimation framework, as detailed below.
2.4. The BrainAGE Estimation Framework
The BrainAGE framework uses relevance vector regression (RVR), a machine learning approach based on pattern recognition. It has been successfully applied in a range of studies (Franke et al., 2010, 2012, 2013; Gaser et al., 2013; Franke et al., 2014, 2015) and was originally de- veloped to model the healthy aging patterns of the brain, as detailed elsewhere (Franke et al., 2010, 2012). Briefly, the initial model has been trained using the preprocessed gray matter segments in a sample of more than 650 subjects, aged between 19 and 86 years. Information on the most important brain regions used by the RVR for estimating the individual brain ages has been provided in the accompanying methods paper (Franke et al., 2010). When applied to new brain scans, specifically the preprocessed gray matter segments extracted from T1-weighted images, the trained algorithm generates an estimated brain age. The concept of the brain age estimation is illustrated in Fig. 1. The difference between estimated age and true chronological age yields the so-called brain age gap estimate (BrainAGE). The absolute BrainAGE index is large if estimated brain age and chronological age are far apart; it is small if both values are close together. The BrainAGE index is negative if a brain is estimated younger than its chronological age (decelerated brain aging); it is positive if a brain is estimated older than its chronological age (accelerated brain aging). For example, if the algorithm computes a BrainAGE index of +5 for the brain of a 50-year-old, this individual shows the typical aging pattern of a 55-year-old. Conversely, if the algorithm computes a BrainAGE index of −5 for the brain of a 50-year-old, this individual shows the typical aging pattern of a 45-year-old. The BrainAGE methodology has been validated across datasets, age ranges and scanner types and was found to be robust and reliable (Franke et al., 2010).
2.5. Main Analyses
First, we computed the BrainAGE index (i.e., the difference between estimated brain age and chronological age) for all 100 subjects, as de- scribed above. Then, the association between BrainAGE, chronological age and meditation was investigated using multiple regression analysis, where chronological age was centered on 50 years. BrainAGE was entered as dependent variable and chronological age and group (meditators/controls) as predictors, while controlling for sex and hand- edness using a factorial design testing all two-way interactions. The model was then reduced to only include significant/influential components. The final model included the following predictors: age, group, sex, handedness, age × group, age × sex, age × handedness,
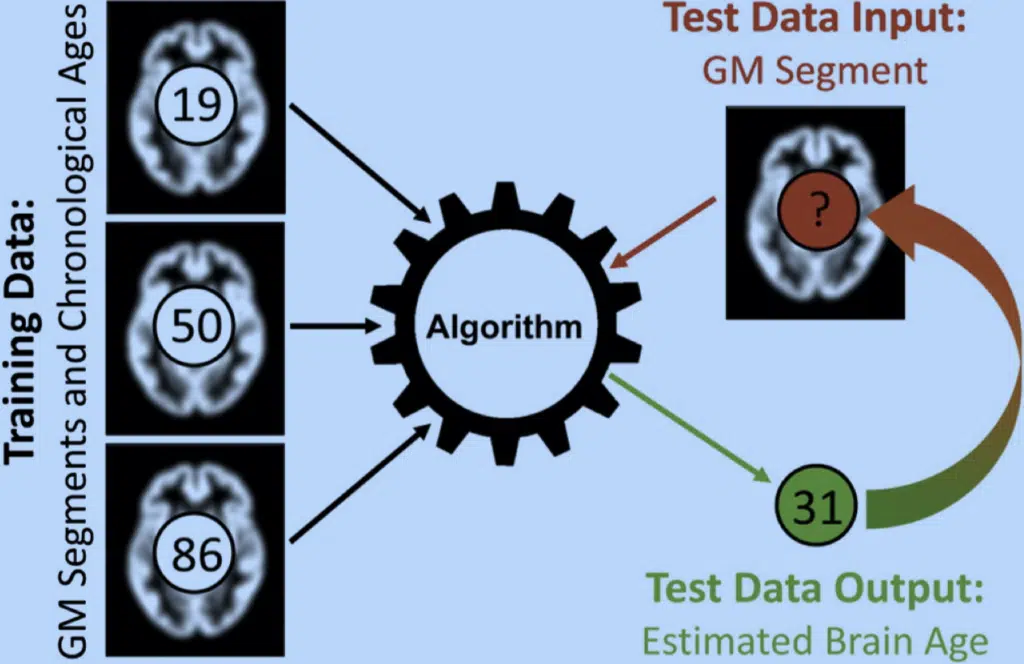
Fig. 1. The brain age estimation concept. First, the algorithm is trained using preprocessed gray matter (GM) segments as well as the individual chronological ages (e.g., 19, 50, and 86 years) from a large training sample. Then, feeding the trained algorithm with the preprocessed GM segments (input data) from a new test sample will provide the estimated brain ages for each subject (e.g., 31 years) as the desired output data.
and group × handedness. For all analyses, alpha was set at 0.05 (two- tailed). Preceding this main analysis, we computed the Pearson correla- tion coefficients between estimated brain age and chronological age within meditators and within controls providing a validation check for the individually obtained brain age estimates: The estimated age was significantly correlated with chronological age, both within controls (r = 0.883; p b 0.001) and within meditators (r = 0.866; p b 0.001). Fig. 2 (top panel) shows the respective scatterplots illustrating the dis- tribution of the brain age estimates within controls and meditators.
2.6. Exploratory Analyses
For exploratory purposes, we also tested for significant correlations between BrainAGE and the number of meditation practice years. Since the current study used a sample with a wide age range (24–77 years) with 62% of all meditators being ≥50 years old, we repeated the afore- mentioned correlation analysis while statistically removing the variance associated with chronological age. In addition, we performed another set of analyses comparing BrainAGE between less experienced and more experienced meditators using a median split (at 16 years of prac- tice), while keeping both groups similar in age on average. Since only a very small subsample of the older practitioners ≥ 50 years (n = 31) had less than 16 years of practice (n = 5), we also repeated this analysis ap- plying the split at 20 years, which resulted in a slightly larger subsample (n = 10). However, none of these four exploratory analyses resulted in any significant effects.
3. Results
Our analyses revealed a main effect of Group (Beta = −7.534, p = 0.047) indicating that, at age 50, brains of meditators were estimated on average to be 7.5 years younger than brains of controls. In addition, there was a main effect of Sex (Beta = 3.398, p = 0.009) indicating that female brains were estimated on average to be 3.4 years younger than male brains. Furthermore, while the main effect of Age was not significant (Beta = 0.404, n.s.), we observed a significant age-by- group interaction (Beta = −0.205, p = 0.050). Follow-up stratified analyses by Group revealed that BrainAGE did not vary as a function of chronological age in controls (Beta = 0.033, n.s.). In contrast, a signif- icant association between BrainAGE and chronological age was detected in meditators (Beta = − 0.144, p = 0.045) indicating that for every one year increase in chronological age over 50 years, there was a decrease of 1 month and 22 days in the estimated brain age. Fig. 2 (bottom panel)
Fig. 2. Group-specific links between age-related measures. Scatterplots and regression lines were generated separately for controls (circles) and meditators (triangles). Top panel: The X-axes display the chronological age; the Y-axes display the estimated brain age (the dashed lines show the confidence interval). Bottom Panel: The X-axes display the chronological age; the Y-axes display the BrainAGE index (negative values indicate that participants’ brains were estimated as younger than their chronological age, positive values indicate that participants’ brains were estimated as older).
shows the respective scatterplots illustrating the distribution of BrainAGE within controls and meditators.
4. Discussion
We applied a recently developed and validated high-dimensional pattern recognition approach estimating the brain ages of 50 long- term meditators and 50 controls based on their structural brain images. Although both groups were perfectly matched for age, our analyses re- vealed significantly lower brain ages in meditators (i.e., more than seven years younger at age fifty) than in controls. These findings seem to imply a decreased brain aging in meditation practitioners.
4.1. Correspondence With Previous Findings
The assumption of younger brains and/or slower brain aging in meditation practitioners is in close agreement with previous findings by other investigators as well as by our own group. For example, a study by Lazar et al. (2005) focusing on cortical thickness in two predetermined brain areas (right insula and right frontal cortex) ex- posed an age-by-group interaction for the right frontal region, with sig- nificant age-related decreases in the control group, but non-significant decreases in the meditation group. Moreover, it was reported that “the average cortical thickness of the 40–50-year-old meditation partici- pants was similar to the average thickness of the 20–30-year-old med- itators and controls” (Lazar et al., 2005).
Another study by Pagnoni and Cekic (2007) focused on whole-brain as well as voxel-wise gray matter. With respect to whole-brain gray matter, there was a trend for a significant age-by-group interaction “with an estimated rate of change of −4.7 ml/year for the control group versus +1.8 ml/year for the meditators group” (Pagnoni and Cekic, 2007). Moreover, the study exposed marginally significant age- related decreases in the control group contrasting increases in the med- itation group (although the latter was non-significant). With respect to voxel-wise gray matter, the same study revealed a significant age-by- group interaction within the left putamen; follow-up analyses stratified by group confirmed the observed whole-brain pattern also within the putamen, with significant negative correlations in controls and a trend for significant positive correlations in meditators (Pagnoni and Cekic, 2007).
These two earlier studies pioneering this field of research preceded our own, more recent aging-related analyses focusing on fractional an- isotropy in twenty fiber tracts (Luders et al., 2011), whole-brain and voxel-wise gray matter throughout the entire brain (Luders et al., 2014), as well as subiculum-specific gray matter volumes (Kurth et al., 2015a). These three latter studies were based on identical or overlap- ping samples with the current study. Therefore, caution is warranted to not overly interpret these outcomes in relation to our present find- ings. Nevertheless, given that each of the studies focused on an entirely different brain measure, it seems at least worth mentioning that study outcomes unanimously support the notion that meditation is associated with reduced age-related brain atrophy.
All of these aforementioned studies (Lazar et al., 2005; Pagnoni and Cekic, 2007; Luders et al., 2011, 2014; Kurth et al., 2015a) were based on standard correlation analyses using chronological age and various brain attributes (e.g., voxel-wise gray matter, point-wise cortical thickness, or tract-specific fractional anisotropy) as variables of interest. In contrast, the current study utilized an established machine learning framework capturing the complex and multidimensional aging pattern across the entire brain and effectively translating it into one single score. Using this highly informative index as dependent variable, we did not only compare the estimated brain ages between groups (meditators vs. controls), but also examined whether associations between chronolog- ical age and estimated brain age vary over time. Indeed, our analyses revealed an intriguing effect confined to the group of meditators: for every year after the age of fifty, meditators’ brains seem to become an additional 1 month and 22 days younger than their chronological age.
As reviewed elsewhere (Fox et al., 2014), a few studies have investi- gated the longitudinal effects of meditation on brain structure (albeit only over short follow-ups) revealing actual meditation-induced increases of brain tissue. Our findings are consistent with those reports in that if meditation practice is associated with increased brain volumes, it may lead to seemingly younger brains. In other words, one of the reasons that the applied high-resolution pattern-based algorithm re- vealed lower brain ages in meditators may be that tissue concentrations in specific brain regions (i.e., those deemed as age-relevant when train- ing the algorithm) were higher than what chronological age would dictate.
4.2. Possible Underlying Mechanisms
An unresolved question relates to the nature of mechanisms under- lying a possible age-defying effect associated with mindfulness practice. As discussed in detail elsewhere (Luders, 2014; Luders et al., 2014; Kurth et al., 2015a), different mechanisms seem plausible, either alone or in conjunction with each other. For example, it is possible that engag- ing in intense mental processes (e.g., as occurring during meditation) induces dendritogenesis, synaptogenesis, myelinogenesis and/or even neurogenesis in the adult brain (Eriksson et al., 1998). Similar events on the micro-anatomical level might occur when, as a consequence of meditation, there is a regular and stronger than normal activation of various neurochemical systems or brain structures associated with pos- itive mood, attention, memory, self-reflection, etc. In all cases, medita- tion could lead to relative increases of tissue in specific brain regions over time. In parallel, it is possible that meditation slows down or counteracts brain tissue loss by setting off biological processes with downstream effects such as the inhibition of cellular apoptosis or the stimulation of cellular preservation/restoration by affecting specific pathways. For example, as recently discussed (Innes and Selfe, 2014), meditation might reduce activation and reactivity of the sympathoadrenal system and the HPA axis, shift the autonomic nervous system from primarily sympathetic to parasympathetic, and stimulate telomerase activity while decreasing pro-inflammatory processes and the production of reactive oxygen species, altogether positively affect- ing resistance to neurodegeneration. If mechanisms underlying tissue gain and tissue conservation work effectively together, the resulting net effect might be sufficiently strong, so that there will be an increase or less decrease in brain tissue globally and/or locally over time. This might also partly explain the aforementioned observation by Pagnoni and Cekic (2007) that the putamen’s gray matter showed a trend for a positive correlation with age in meditators (while showing a significant negative correlation in controls).
To summarize, normal aging is associated with a significant loss of brain tissue. Thus, meditation-induced tissue gain and/or meditation- induced tissue conservation might ultimately manifest as a reduced brain age (i.e., cerebral measures characteristic of younger brains) as we have reported when comparing the mean brain age index between meditators and controls at age fifty. If meditation-related benefits for the human brain accumulate over time, additive age-defying effects might become evident in later years of life, as also currently observed in our sample of long-term meditation practitioners after the age of fifty (i.e., the higher the chronological age, the larger the discrepancy be- tween chronological age and estimated brain age towards relatively younger brains in older practitioners).
4.3. An Intriguing Sex Effect
Last but not least, while it was not the main focus of our study but merely a by-product of including sex as a covariate and testing for all two-way interactions, we observed striking differences between men and women with respect to their estimated brain ages. More specifically, female brains at age 50 were estimated more than three years younger than male brains. This sex effect was evident in the combined sample (i.e., independent of meditation status) and seems to be in agreement with previous epidemiological data indicating that women not only tend to live longer but also show somewhat less age-related brain atrophy (Coffey et al., 1998; Xu et al., 2000; Raz et al., 2004).
4.4. Strengths, Limitations and Future Studies
This study had a number of limitations but also several strengths. Due to its cross-sectional nature, causality of the reported effects cannot be resolved. It is possible, for example, that brains of those who engage in meditative practices are inherently different from those who do not. The level of education might be another confound (Steffener and Stern, 2012; Mortby et al., 2014) and could have driven, at least partly, the out- comes of the current study: While meditators and controls were some- what comparable in that 90% of the meditators and 96% of the controls had at least some college experience, the proportion of individuals with Master’s degrees was higher among meditators than controls (46% ver- sus 20%). At the same time though, the proportion of individuals with only high school degrees was lower among controls than meditators (4% versus 10%). It is also possible that certain lifestyle factors associated with meditation underlie our findings. For example, if meditators differ from non-meditators with respect to their diet, physical exercise, alco- hol consumption, smoking habits, or general health status – all of which have been reported to significantly affect aging processes in the human brain (Iso et al., 2004; Erickson et al., 2011; Duriez et al., 2014; Cherbuin et al., 2015; Jacka et al., 2015) – some of the currently reported effect (i.e., younger brains in meditation practitioners) may potentially be due to group differences in those variables.
Future studies will enhance this field of research by capturing such dis- criminating features between meditators and controls (aside from the ac- tual practice of meditation) and either statistically remove the impact of potential moderator variables or use them as inclusion/exclusion or matching criteria during subject selection. Moreover, longer term longitu- dinal follow-ups will be necessary to resolve the nature/nurture debate and to elucidate the role meditation may play in brain aging overall. Partic- ular strengths of this study are the methodologically solid approach, the large sample size, the wide age range, and the fact that meditators and controls were closely matched on sex and age and also came from the same population. In addition to characterizing samples with respect to confounding variables and adopting longitudinal designs, future studies should further scrutinize the underlying biological mechanisms which un- derpin a beneficial effect of meditation, specifically with regard to HPA axis modulation, neurogenesis, apoptosis, inflammation and oxidative stress.
Disclosure Statement
There are no actual or potential conflicts of interest. Acknowledgments
We wish to thank all meditators for their participation in our study and we are grateful to Trent Thixton who assisted with the acquisition of the image data. NC is funded by the Australian Research Council fu- ture fellowship number 120100227.
References
Ashburner, J., Friston, K.J., 2005. Unified segmentation. NeuroImage 26, 839–851. Cherbuin, N., Sargent-Cox, K., Fraser, M., Sachdev, P., Anstey, K.J., 2015. Being overweight is
associated with hippocampal atrophy: the PATH through life study. Int. J. Obes.
Coffey, C.E., Lucke, J.F., Saxton, J.A., Ratcliff, G., Unitas, L.J., Billig, B., Bryan, R.N., 1998. Sex differences in brain aging: a quantitative magnetic resonance imaging study. Arch.
Neurol. 55, 169–179.
Cuadra, M.B., Cammoun, L., Butz, T., Cuisenaire, O., Thiran, J.P., 2005. Comparison and val- idation of tissue modelization and statistical classification methods in T1-weighted MR brain images. IEEE Trans. Med. Imaging 24, 1548–1565.
Duriez, Q., Crivello, F., Mazoyer, B., 2014. Sex-related and tissue-specific effects of tobacco smoking on brain atrophy: assessment in a large longitudinal cohort of healthy elder- ly. Front. Aging Neurosci. 6, 299.
Erickson, K.I., Voss, M.W., Prakash, R.S., Basak, C., Szabo, A., Chaddock, L., Kim, J.S., Heo, S., Alves, H., White, S.M., Wojcicki, T.R., Mailey, E., Vieira, V.J., Martin, S.A., Pence, B.D., Woods, J.A., McAuley, E., Kramer, A.F., 2011. Exercise training increases size of hippo- campus and improves memory. Proc. Natl. Acad. Sci. U. S. A. 108, 3017–3022.
Eriksson, P.S., Perfilieva, E., Bjork-Eriksson, T., Alborn, A.M., Nordborg, C., Peterson, D.A., Gage, F.H., 1998. Neurogenesis in the adult human hippocampus. Nat. Med. 4, 1313–1317. Fox, K.C., Nijeboer, S., Dixon, M.L., Floman, J.L., Ellamil, M., Rumak, S.P., Sedlmeier, P.,
Christoff, K., 2014. Is meditation associated with altered brain structure? A systematic review and meta-analysis of morphometric neuroimaging in meditation practi- tioners. Neurosci. Biobehav. Rev. 43C, 48–73.
Franke, K., Ziegler, G., Kloppel, S., Gaser, C., 2010. Estimating the age of healthy subjects from T1-weighted MRI scans using kernel methods: exploring the influence of vari- ous parameters. NeuroImage 50, 883–892.
Franke, K., Luders, E., May, A., Wilke, M., Gaser, C., 2012. Brain maturation: predicting individual BrainAGE in children and adolescents using structural MRI. NeuroImage 63, 1305–1312.
Franke, K., Gaser, C., Manor, B., Novak, V., 2013. Advanced BrainAGE in older adults with type 2 diabetes mellitus. Front. Aging Neurosci. 5, 90.
Franke, K., Ristow, M., Gaser, C., 2014. Gender-specific impact of personal health param- eters on individual brain aging in cognitively unimpaired elderly subjects. Front. Aging Neurosci. 6, 94.
Franke, K., Hagemann, G., Schleussner, E., Gaser, C., 2015. Changes of individual BrainAGE during the course of the menstrual cycle. NeuroImage 115, 1–6.
Fraser, M.A., Shaw, M.E., Cherbuin, N., 2015. A systematic review and meta-analysis of longitudinal hippocampal atrophy in healthy human ageing. NeuroImage.
Gaser, C., Franke, K., Kloppel, S., Koutsouleris, N., Sauer, H., 2013. BrainAGE in mild cogni- tive impaired patients: predicting the conversion to Alzheimer’s disease. PLoS One 8, e67346.
Innes, K.E., Selfe, T.K., 2014. Meditation as a therapeutic intervention for adults at risk for Alzheimer’s disease — potential benefits and underlying mechanisms. Front. Psychi- atry 5, 40.
Iso, H., Baba, S., Mannami, T., Sasaki, S., Okada, K., Konishi, M., Tsugane, S., 2004. Alcohol consumption and risk of stroke among middle-aged men: the JPHC Study Cohort I. Stroke 35, 1124–1129.
Jacka, F.N., Cherbuin, N., Anstey, K.J., Sachdev, P., Butterworth, P., 2015. Western diet is associated with a smaller hippocampus: a longitudinal investigation. BMC Med. 13, 215.
Kurth, F., Cherbuin, N., Luders, E., 2015a. Reduced age-related degeneration of the hippo- campal subiculum in long-term meditators. Psychiatry Res. 232, 214–218.
Kurth, F., MacKenzie-Graham, A., Toga, A.W., Luders, E., 2015b. Shifting brain asymmetry: the link between meditation and structural lateralization. Soc. Cogn. Affect. Neurosci. 10, 55–61.
Lazar, S.W., Kerr, C.E., Wasserman, R.H., Gray, J.R., Greve, D.N., Treadway, M.T., McGarvey, M., Quinn, B.T., Dusek, J.A., Benson, H., Rauch, S.L., Moore, C.I., Fischl, B., 2005. Medita- tion experience is associated with increased cortical thickness. Neuroreport 16, 1893–1897.
Luders, E., 2014. Exploring age-related brain degeneration in meditation practitioners. Ann. N. Y. Acad. Sci. 1307, 82–88.
Luders, E., Toga, A.W., Lepore, N., Gaser, C., 2009. The underlying anatomical correlates of long-term meditation: larger hippocampal and frontal volumes of gray matter. NeuroImage 45, 672–678.
Luders, E., Clark, K., Narr, K.L., Toga, A.W., 2011. Enhanced brain connectivity in long-term meditation practitioners. NeuroImage 57, 1308–1316.
Luders, E., Kurth, F., Mayer, E.A., Toga, A.W., Narr, K.L., Gaser, C., 2012a. The unique brain anatomy of meditation practitioners: alterations in cortical gyrification. Front. Hum. Neurosci. 6, 34.
Luders, E., Phillips, O.R., Clark, K., Kurth, F., Toga, A.W., Narr, K.L., 2012b. Bridging the hemispheres in meditation: thicker callosal regions and enhanced fractional anisotro- py (FA) in long-term practitioners. NeuroImage 61, 181–187.
Luders, E., Kurth, F., Toga, A.W., Narr, K.L., Gaser, C., 2013a. Meditation effects within the hippocampal complex revealed by voxel-based morphometry and cytoarchitectonic probabilistic mapping. Front. Psychol. 4, 398.
Luders, E., Thompson, P.M., Kurth, F., Hong, J.Y., Phillips, O.R., Wang, Y., Gutman, B.A., Chou, Y.Y., Narr, K.L., Toga, A.W., 2013b. Global and regional alterations of hippocampal anato- my in long-term meditation practitioners. Hum. Brain Mapp. 34, 3369–3375.
Luders, E., Cherbuin, N., Kurth, F., 2014. Forever young(er): potential age-defying effects of long-term meditation on gray matter atrophy. Front. Psychol. 5, 1551.
Luders, E., Thompson, P.M., Kurth, F., 2015. Larger hippocampal dimensions in meditation practitioners: differential effects in women and men. Front. Psychol. 6, 186.
Mortby, M.E., Burns, R., Janke, A.L., Sachdev, P.S., Anstey, K.J., Cherbuin, N., 2014. Relating education, brain structure, and cognition: the role of cardiovascular disease risk factors. BioMed Res. Int. 2014, 271487.
Pagnoni, G., Cekic, M., 2007. Age effects on gray matter volume and attentional perfor- mance in Zen meditation. Neurobiol. Aging 28, 1623–1627.
Pfefferbaum, A., Rohlfing, T., Rosenbloom, M.J., Chu, W., Colrain, I.M., Sullivan, E.V., 2013. Variation in longitudinal trajectories of regional brain volumes of healthy men and women (ages 10 to 85 years) measured with atlas-based parcellation of MRI. NeuroImage 65, 176–193.
Rajapakse, J.C., Giedd, J.N., Rapoport, J.L., 1997. Statistical approach to segmentation of single-channel cerebral MR images. IEEE Trans. Med. Imaging 16, 176–186.
Raz, N., Gunning-Dixon, F., Head, D., Rodrigue, K.M., Williamson, A., Acker, J.D., 2004.
Aging, sexual dimorphism, and hemispheric asymmetry of the cerebral cortex: repli-
cability of regional differences in volume. Neurobiol. Aging 25, 377–396.
Raz, N., Ghisletta, P., Rodrigue, K.M., Kennedy, K.M., Lindenberger, U., 2010. Trajectories of brain aging in middle-aged and older adults: regional and individual differences.
NeuroImage 51, 501–511.
Steffener, J., Stern, Y., 2012. Exploring the neural basis of cognitive reserve in aging. Biochim. Biophys. Acta 1822, 467–473.
Tohka, J., Zijdenbos, A., Evans, A., 2004. Fast and robust parameter estimation for statisti- cal partial volume models in brain MRI. NeuroImage 23, 84–97.
Xu, J., Kobayashi, S., Yamaguchi, S., Iijima, K., Okada, K., Yamashita, K., 2000. Gender effects on age-related changes in brain structure. AJNR Am. J. Neuroradiol. 21, 112–118.